A day in the life of a chemist
Enhancing the drug discovery ideation process
Every new small molecule drug starts with an idea. As significant time and resources are required to take a drug to market, the rationale behind each new molecule design must be well-defined prior to committing to synthesis and analysis.
Torx® enables chemists to keep a comprehensive record of all known information surrounding a project, so that new ideas can be recorded and evaluated in seconds. In this blog installment, we explore the different ways that Torx transforms the ideation process, so that you can be sure you’re only committing those designs which give you the highest chance of success.
Record a new molecule design anytime, anywhere
New ideas don’t always come to mind at the most convenient times, such as when you’re at your desk with your notebook in hand. Whether you’re in the lab, at home, or on your travels, you can add new molecules instantly into Torx Design, while the thought is still fresh in your mind.
Validate your new molecule designs against existing knowledge
Now that you’ve made a record of your idea(s), you may wish to first validate them yourself as much as you can, before sharing with the rest of the team. With a fully customizable GUI, Torx Design delivers the most relevant, up-to-date information and metadata in one single, easily-accessible place as you edit your molecule in real time. This includes highlighting similar compounds in the company portfolio, that have either been attempted previously or are currently in progress. Knowing which molecules have been successful (and just as importantly, which molecules have been unsuccessful), ensures that you waste no unnecessary time or resources in the lab.
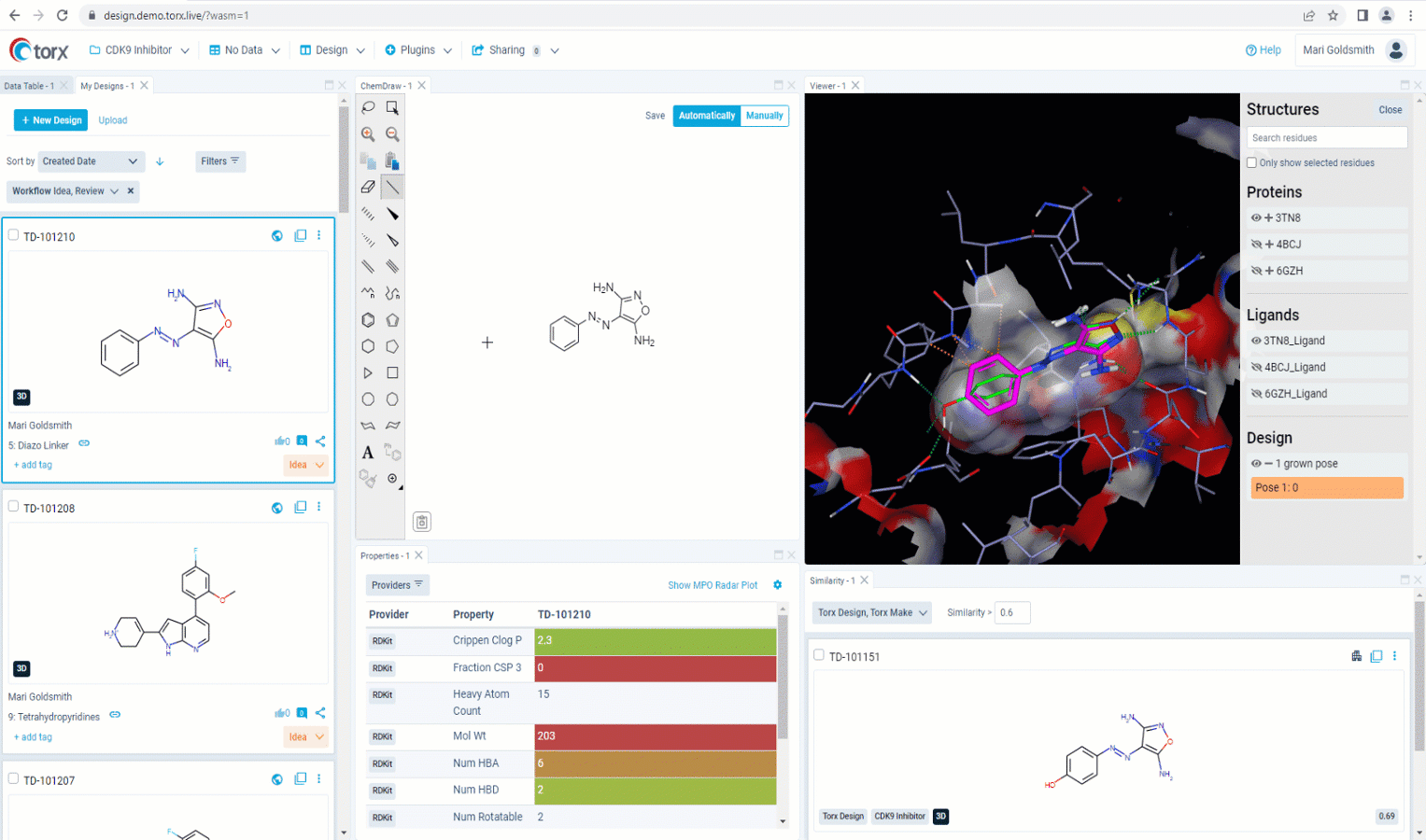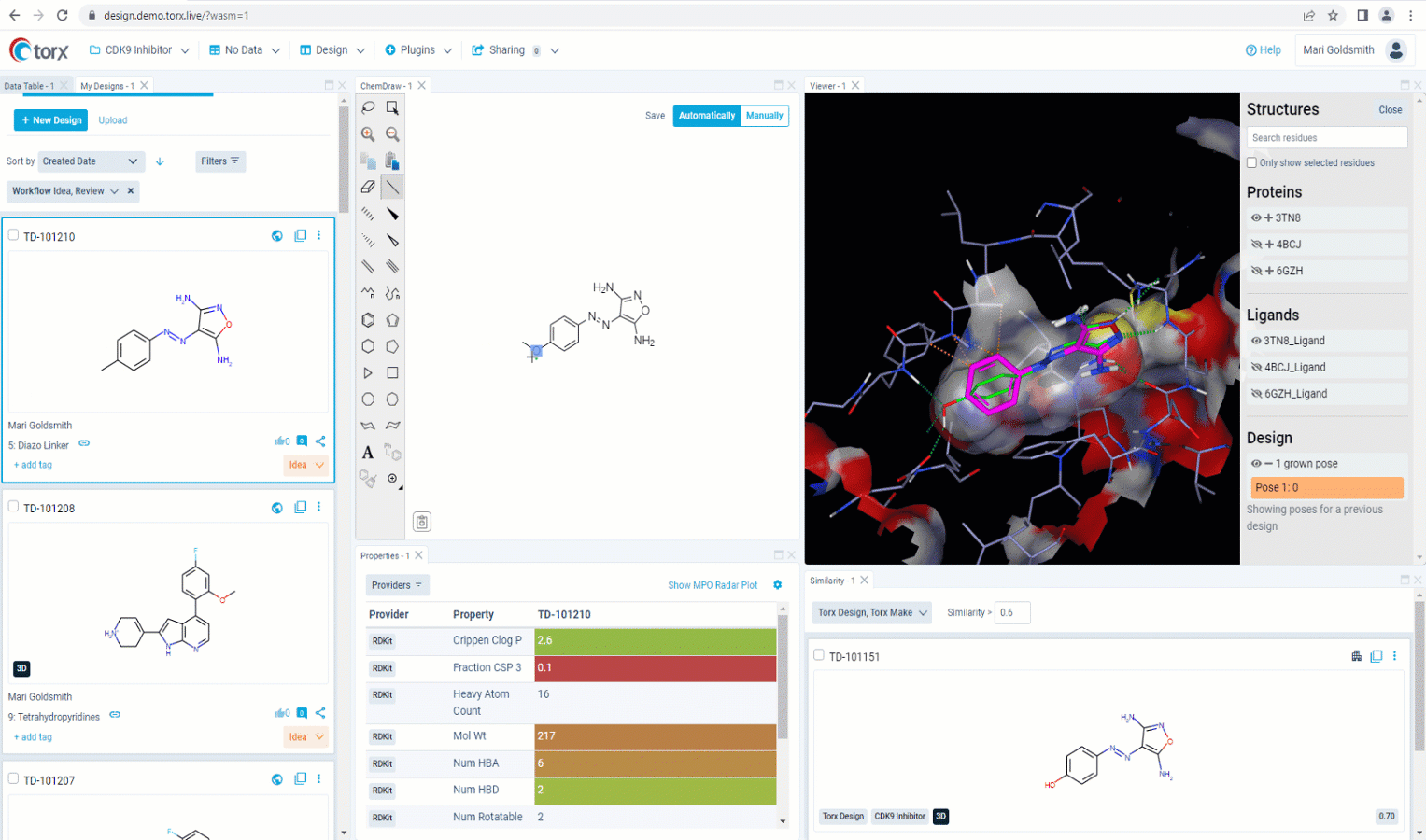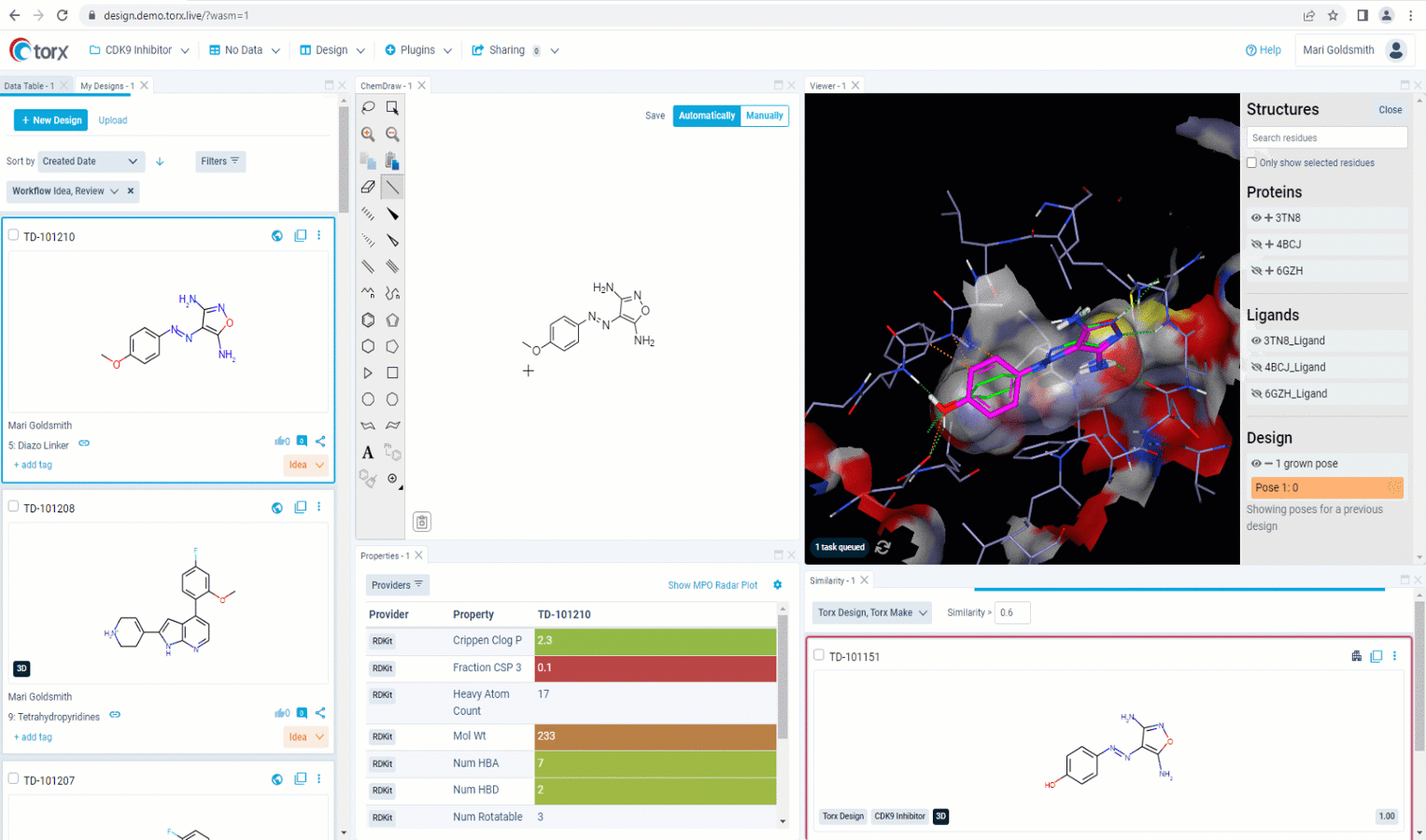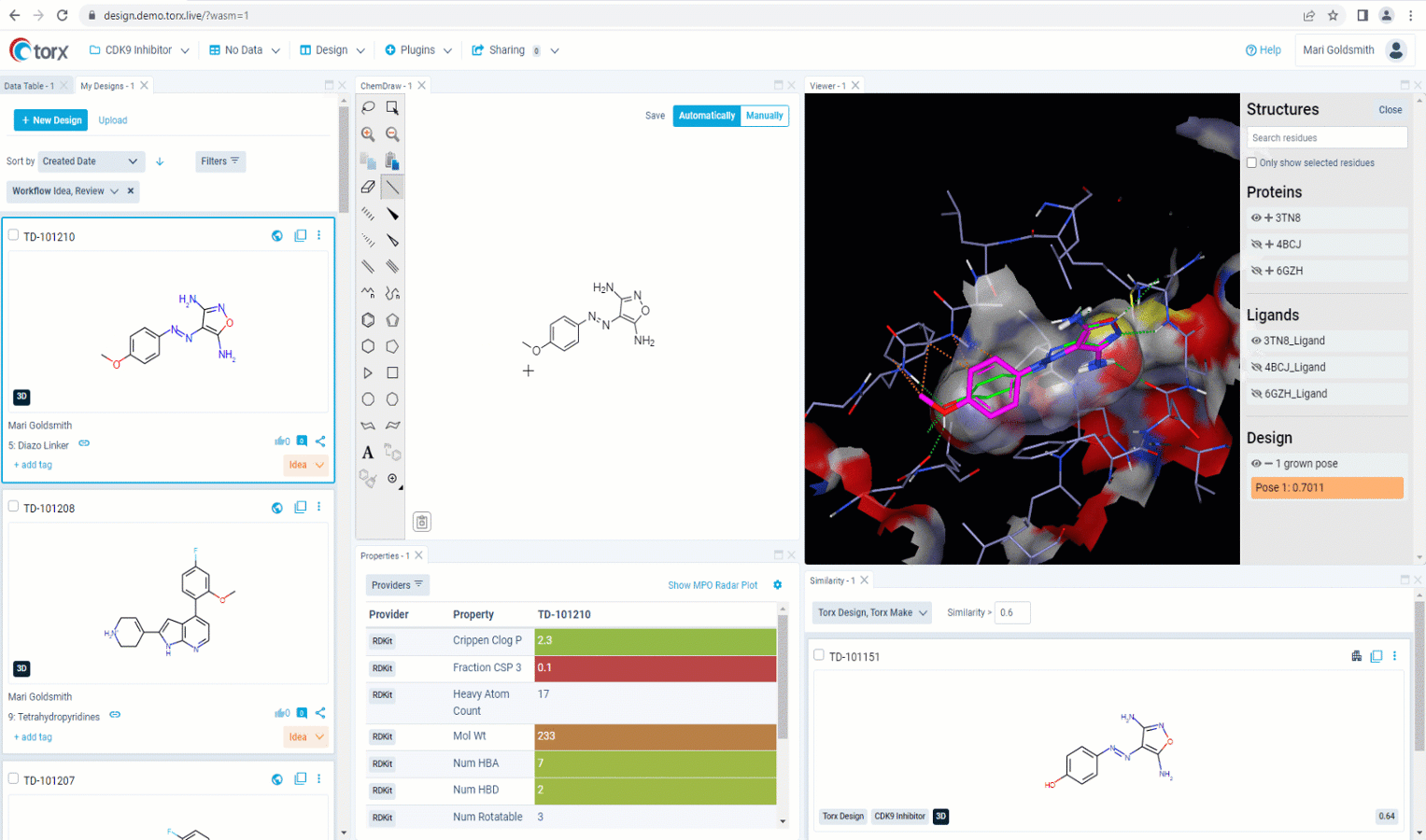
Figure 2. See relevant information highlighted in real time as you sketch new molecules in Torx Design, including similar molecules that have been previously attempted (in red).
Within each organization, is a legacy of information spanning years. So that you can truly benefit from this wealth of information, Torx ensures that all existing knowledge is captured securely in a single web browser, even after team members have moved on. This information is readily accessible at your fingertips, as opposed to the traditional time-consuming approach of manually trawling through libraries and databases.
Work collaboratively: Share ideas and receive instant feedback from colleagues
Drug discovery is a highly collaborative process. Each interdisciplinary team features specialists in medicinal and computational chemistry, biology and pharmacology, all of who will advise on the direction of the project. Getting insight from the right people in a timely manner is key to research productivity. However, balancing multiple schedules to plan review meetings can be a frustrating task. Sharing ideas via email and waiting for individual feedback can be time consuming and delay progress.
Torx transforms the way that you collaborate with colleagues. New designs are shared in an instant to team members so they can review in the context of all available information. Visible feedback can be given quickly in the form of likes, comments or tags.
We understand that some discussions require further time and input. In these cases, Torx live shared sessions enable you to collaborative with colleagues in real time even when based across different organizations or locations. As molecules are shared, not screens, there is no need to sanitize your desktop or switch contexts – you retain your working environment throughout.
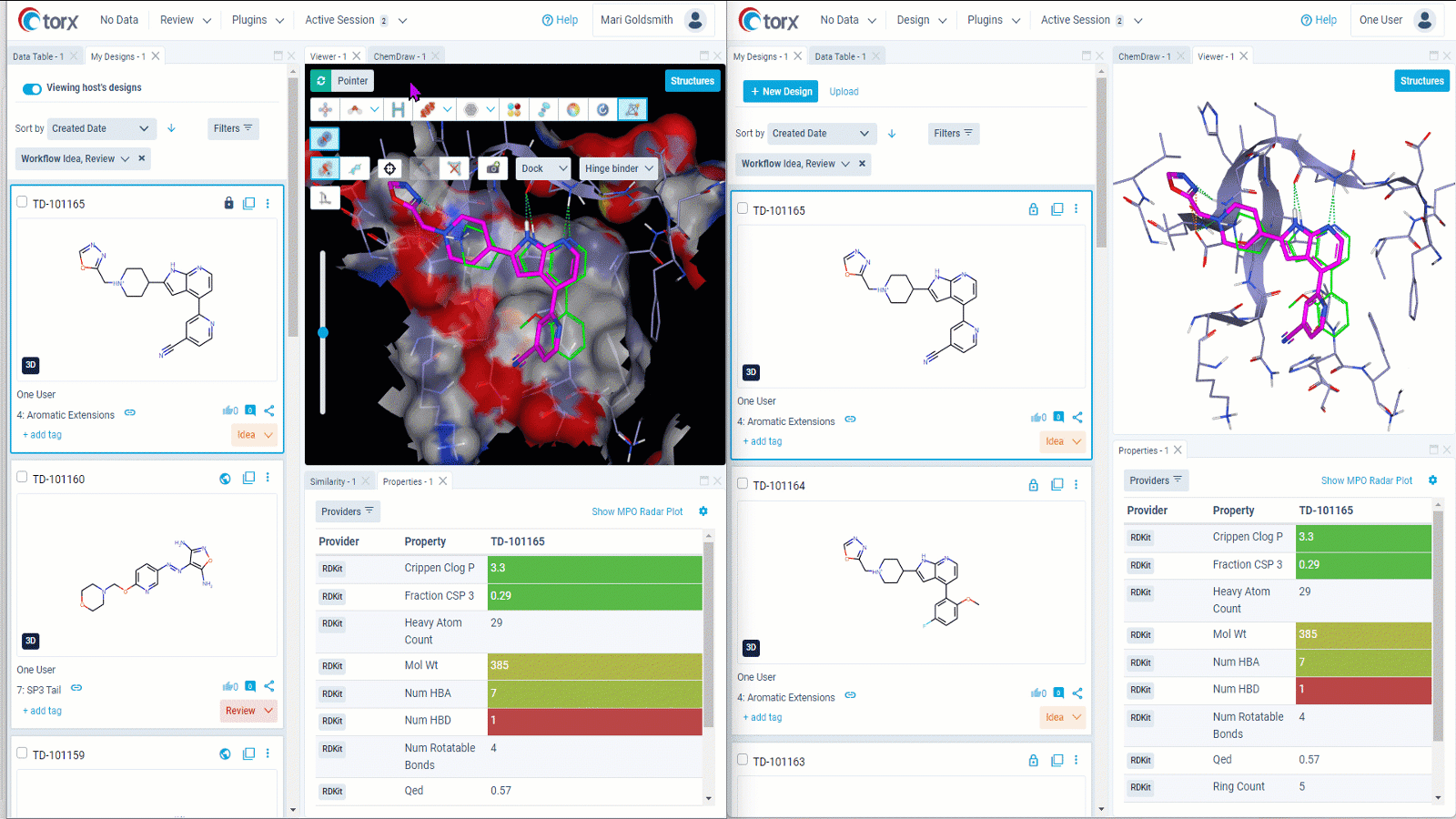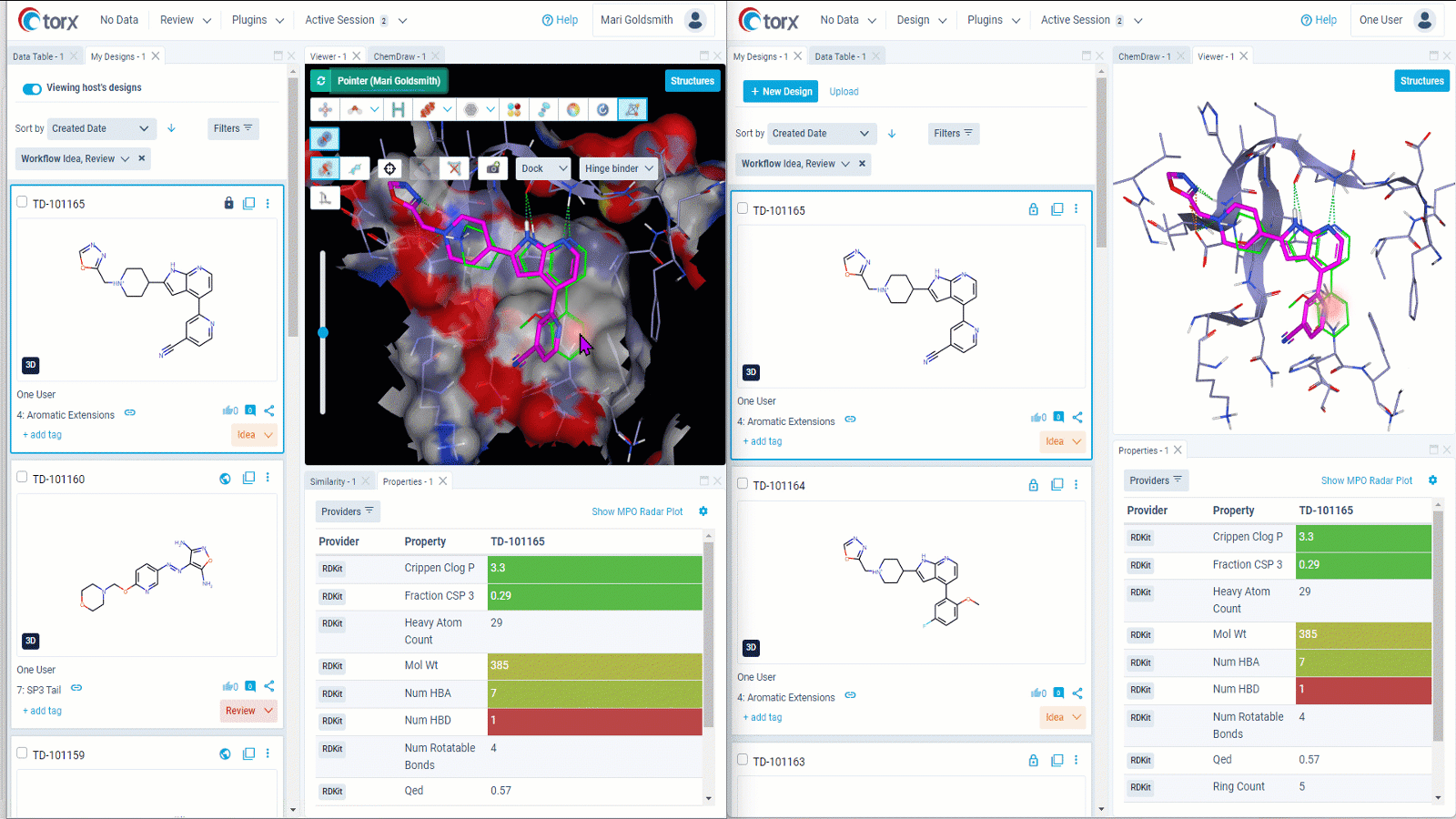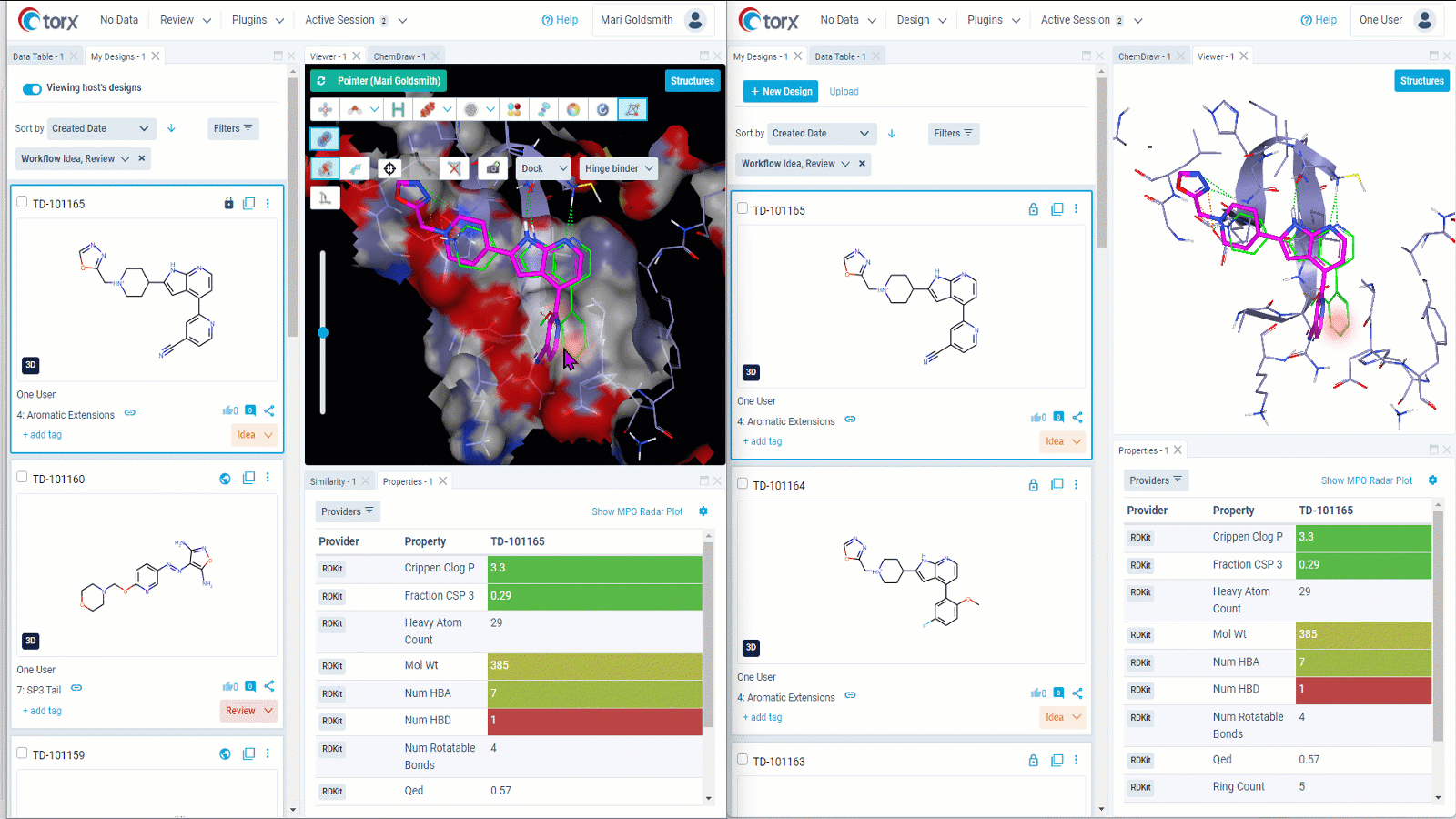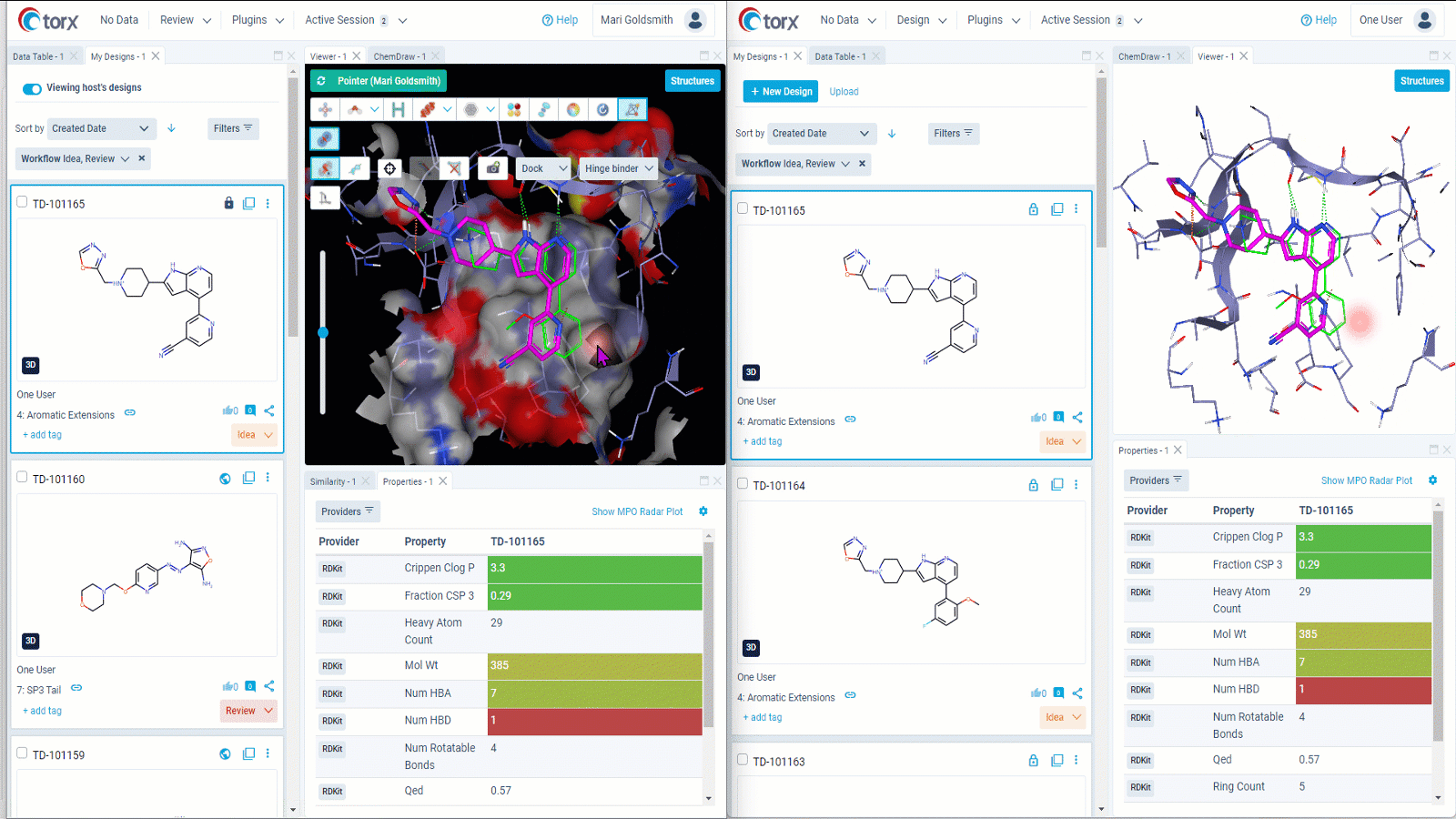
Figure 3: Gain valuable insights on designs in real time by collaborating remotely through Torx live shared sessions. Both users (left and right) retain their own working environment throughout the session.
Track project contributions in a comprehensive audit log
Knowing how each individual has contributed to a successful idea is essential when patenting new compounds. This isn’t always straightforward when ideas are recorded unattributed, and knowing who is associated with each compound is lost in the flurry of activity.
The dedicated Audit log in Torx keeps a comprehensive record of each action to ensure all inventors are recognized. At the click of a mouse, you see everyone that has been involved in each new design, and how.
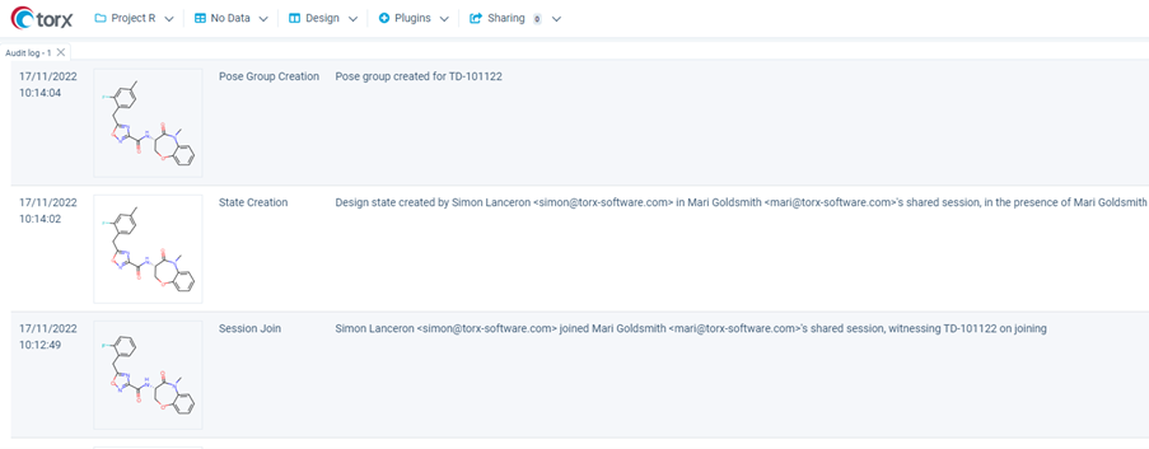
Figure 4. Comprehensively capture all contributions to each new design in the Torx Audit log.
Want to transform your drug discovery ideation process? Try Torx on your project
If you’d like to streamline your processes of recording, validating and attributing new ideas, request an evaluation, or contact us to find out more information.